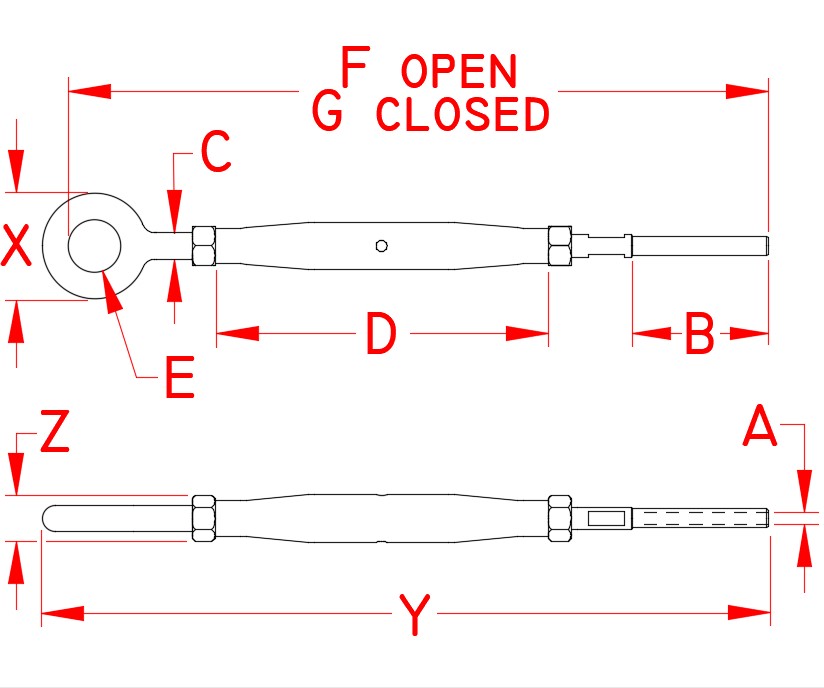
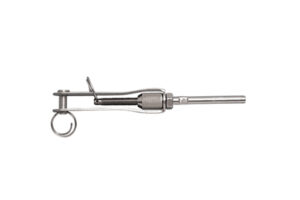
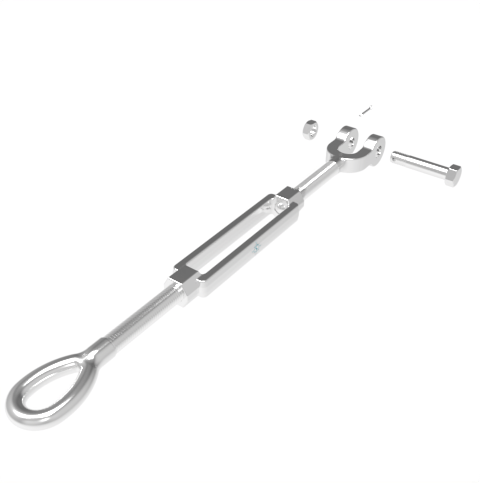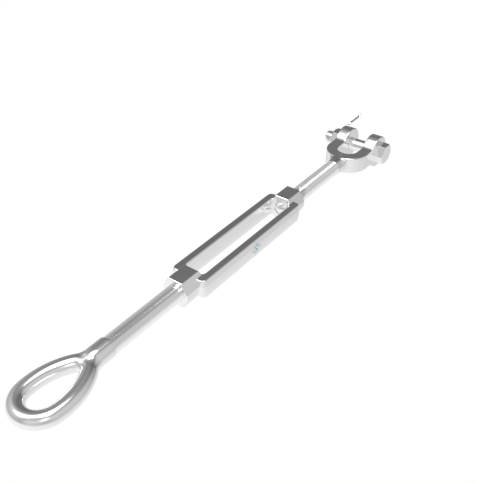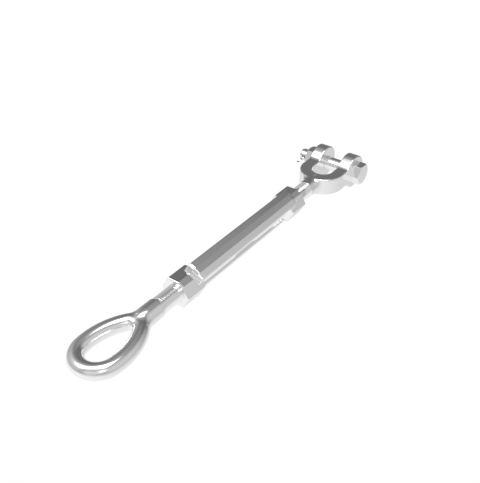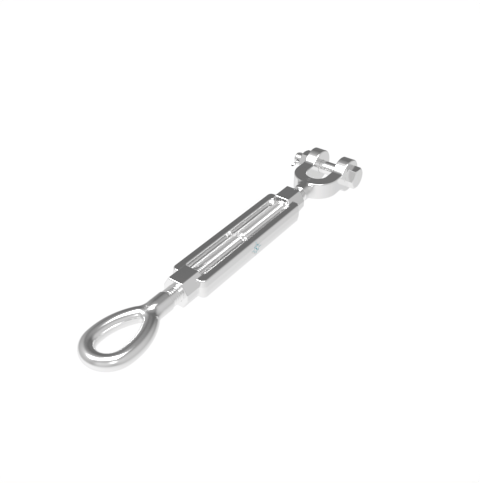
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Material: Chromed Bronze Body and Stainless Ends
Material: Aluminum and Stainless Steel
Material: Aluminum and Stainless Steel
Material: Aluminum and Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Grade: 316-NM Stainless Steel
Material: Bronze Body and Stainless Ends
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Material: Chromed Bronze Body and Stainless Ends
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Material: Chromed Bronze Body & Stainless Ends
Grade: 316 Stainless Steel
Material: Chromed Bronze Body & Stainless Ends
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel
Grade: 316 Stainless Steel